- HOME
- VENUE
- RSVP
- REGISTRY
- CONTACT
- Rainbow srx
- Chozen one spirit filled
- Coreldraw essentials
- Does avast mac security protect from cryptolocker
- Pro video formats adobe
- Knoebels covered bridge 2021
- Splitt steel beam
- The game of life reviews
- Safari storme
- Get jupyterlab extensions
- Polyphonic ringtones
- Part of thebrain that helps keep eyes focused on one thing
- Vega conflict hack tool download free
- Low density lipoprotein
- Ubuntu port forwarding wizard
- Mndot road closures and detours
- Copy log sheet
- World civilization ii online course
- Green hell fishing rod
- Cook county court records
- Pdf merger and splitter online
- Play youtube sonos for mac
- Does zinc rust
- Photomarks app for android
- Docker logs
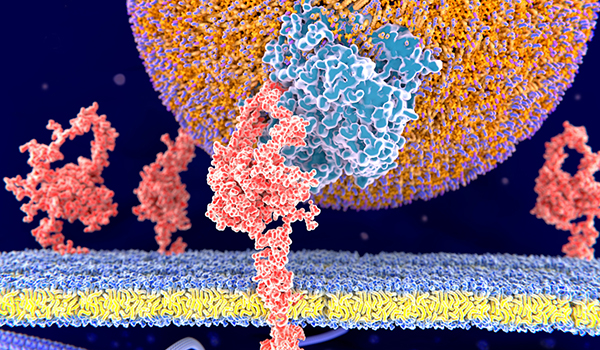
After the first study drug administration, the participant will be observed in the clinic for at least 4 hours post injection before being discharged. On Day 1, all eligible participants will be randomized and receive the first subcutaneous (SC) administration of ALN-PCSSC or placebo. A recommendation may be taken to stop or amend the study at any of these reviews. Thereafter, the DMC will review safety data every 2 months until the end of the trial. The independent Data Monitoring Committee (DMC) will review safety data beginning after the first 40 participants receive the first injection of ALN-PCSSC or placebo and complete the Day 14 follow-up visit. Each participant will receive either one or two injections on Day 1 or a single injection on Day 1 and on Day 90 of blinded ALN-PCSSC or placebo.įormation of anti-drug antibodies (ADA) will be assessed on Day 1 (prior to and 4 hours after the injection) and on Days 30, 60, 90, 120, 150, 180 (Days 150 and 180 only in participants who receive a second dose of study drug), and 210 or until any ADA response becomes negative within the study duration. Treatment allocation will be stratified by country and by current use of statins or other lipid-modifying therapies.
LOW DENSITY LIPOPROTEIN PLUS
Participants will be screened and 480 eligible participants will be randomized: 60 participants per each of six ALN-PCSSC dose groups plus 120 participants total across the placebo groups (20 participants each to match each of the six drug dose groups). Why Should I Register and Submit Results?.Although future studies should be aimed at determining the clinical benefit of reducing sdLDL levels, there is sufficient evidence to warrant consideration of sdLDL measurement in assessing and managing risk of cardiovascular disease.Ĭopyright © 2022 Wolters Kluwer Health, Inc.

Nevertheless, determination of the extent to which sdLDL can preferentially impact ASCVD risk compared with other apoprotein B-containing lipoproteins has been confounded by their metabolic interrelationships and statistical collinearity, as well as differences in analytic procedures and definitions of sdLDL.Ī growing body of data points to sdLDL concentration as a significant determinant of ASCVD risk. larger LDL, including reduced LDL receptor affinity and prolonged plasma residence time as well as greater oxidative susceptibility and affinity for arterial proteoglycans, are consistent with their heightened atherogenic potential. Results of multiple prospective studies have supported earlier evidence that higher levels of sdLDL are significantly associated with greater ASCVD risk, in many cases independent of other lipid and ASCVD risk factors as well as levels of larger LDL particles. This review focuses on the clinical significance of sdLDL measurement. Levels of small, dense low-density lipoprotein (LDL) (sdLDL) particles determined by several analytic procedures have been associated with risk of atherosclerotic cardiovascular disease (ASCVD).